Clinical Trials
Clinical trials are medical research studies that involve people. They may involve patients, healthy participants, or both.
Clinical trials aim to test new treatments or procedures and answer the following questions:
- Is it safe?
- Does it work better than the current standard treatment or procedure?
- Does it have side-effects?
- What is the best dose to give?
- Does it have an impact on quality of life?
What are the different types of clinical trial?
There are two main types of clinical trial: interventional and observational. Interventional studies investigate a new intervention or treatment. Those taking part are often divided into different treatment groups so that the researchers can compare the effects of different treatments. During observational studies, researchers observe and collect information on participants without any intervention.
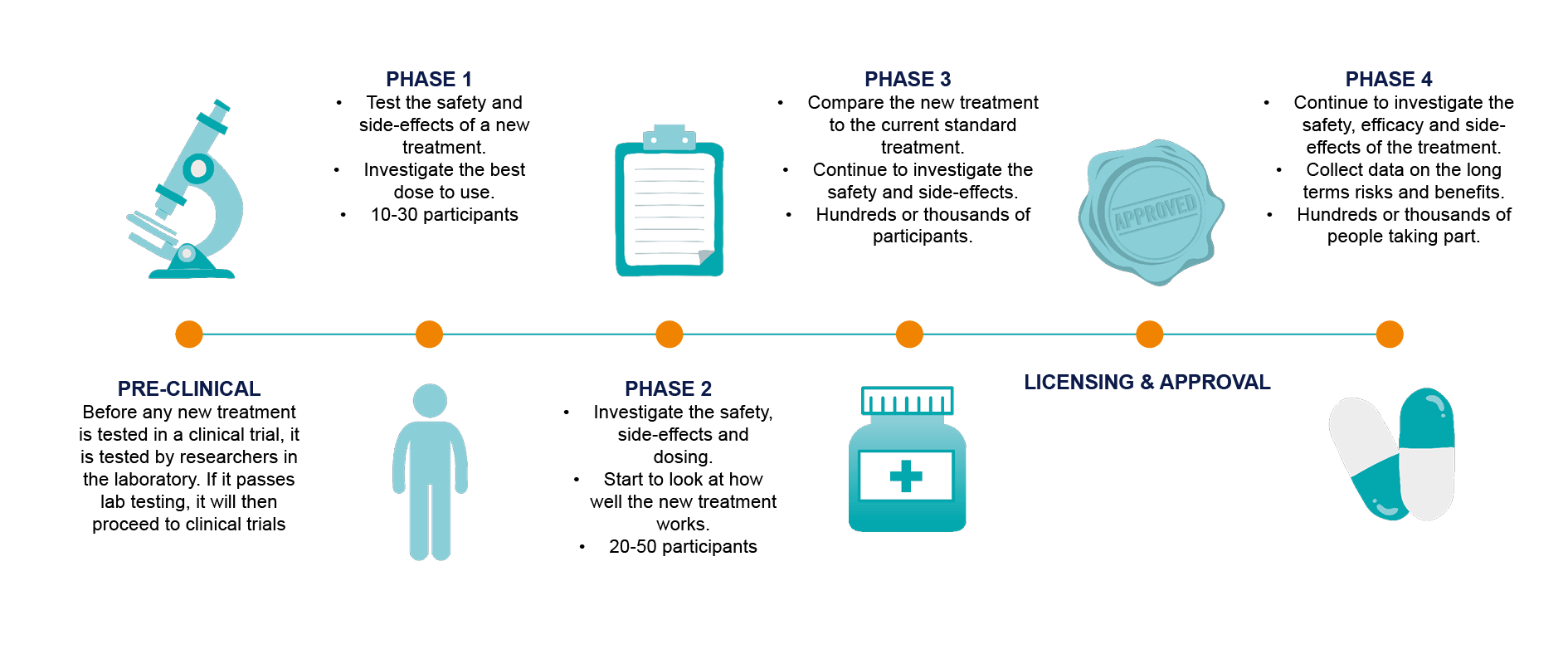
Clinical trials are also divided into four main stages or phases.
What are randomised clinical trials?
Almost all phase 3 trials and some phase 2 trials are randomised. This means that the people taking part in the trial are put into different treatment groups at random. Randomised trials have two or more different treatment groups. There is usually a control group which has the standard treatment. The other group receive the new treatment or procedure that is being tested.
Randomised trials help the scientists to ensure that the results from the trial are reliable and not biased. For example, researchers may put healthier participants into a particular group without realising. These participants are likely to have better outcomes than those who are more unwell so it may make it seem like a particular treatment works better than it does.
What is the placebo effect?
Many people will feel better if they receive a fake treatment, known as a placebo, simply because they believe it will have a positive effect. This is known as the placebo effect. In order to make sure a treatment is actually working, the control group may be given a placebo. Participants would only be given a placebo if it is either given alongside standard treatment or if there is no standard treatment available. If there is no standard treatment, those given the placebo would not receive treatment if they were not taking part in the trial, so the trial does not put them at a disadvantage. It would be unethical to withhold cancer treatment from people who need it so regardless of whether you receive the new treatment, you will always be given the best available treatment and care.
To reduce the placebo effect, trials may be blinded or double-blinded. In blind trials, you do not know whether you have been given the new treatment or not. In double blind trials, neither you nor the doctor administering the treatment knows whether you have been given the new treatment.

Are clinical trials safe?
Before any new treatment is tested in a clinical trial, it is tested by researchers in the laboratory. If it passes lab testing, it will then proceed to clinical trials. Before the trial can begin, the trial plan must be reviewed by doctors and approved by an ethics committee. You will be monitored carefully throughout the study to ensure that any issues are identified as soon as possible.
The researchers should ensure that you understand what the trial involves and the potential side-effects and risks before you take part in the clinical trial. You will be asked to give your consent before you can take part. Should you change your mind, you are able to leave the trial at any time.
Should I take part in a clinical trial?
Deciding whether or not to take part in a clinical trial is very personal and there is no right or wrong answer.
Clinical trials help doctors find new ways to treat prostate cancer which could help others affected by the disease in the future. Taking part in a trial may mean that you receive more regular checks and support and it may also enable you to access and benefit from new cutting-edge treatment.
However, there is no guarantee that you will be given the new treatment as you may be placed in the control group. There is also the possibility that the new treatment does not work better than the current standard treatment and there may be unforeseen side-effects.
How do I join a clinical trial?
If you are interested in joining a clinical trial, the best person to speak to is your doctor or nurse. They will be able to discuss any trials that may be suitable for you to join. If appropriate, they will also be able to contact a doctor involved in the trial and refer you.
You can find clinical trials you may be suitable for using our clinical trial finder on the infopool.
Find clinical trials you may be eligible for using our clinical trial finder